By Jeremy Yeoh
Illustration by Anne-Gaëlle Goubet for SYIS
Have you ever felt like you don’t really fit in a box that was meant to describe you? Do you feel like both a Pisces and a Scorpio at the same time? To make our lives simpler, humans tend to categorize things into neat groups, whether it be ourselves, other humans or immune cells. However, in immunology, classification and categorization of cell identities are getting increasingly difficult as the numbers of different cell types that have been identified are getting out of hand with the advent of new technologies. This is getting even more problematic by the concept of plasticity in biology, creating a professional organizer’s worst nightmare.
Some Immune Cells Are Plastic! (Not The Environmentally Unfriendly Type of Plastic)
The general idea in cell biology is that you start from an omnipotent stem cell, gradually differentiating and gaining different functions until it reaches a terminal permanent state that has defined roles within a system. This is termed as lineage stability. However, what we observe is not that straightforward. Plasticity in immunity is in principle defined as the ability of cells to acquire changes in phenotype and/or function in response to a dynamic environment. This would suggest that plastic cells generally exist more on a spectrum with fluid identities rather than on a binary basis – a landscape of cells instead of defined non-changing personas, complicating the attempted categorization of these cells into tidy little boxes.
It seems that the closer we head towards specialized pathogen-specific immunity, the more we see this phenomenon of plasticity – starting from macrophages and dendritic cells (the bridges to adaptive immunity) to the specific immune arm of the adaptive immune system, lymphocytes. As the immune system gets more specialized and specific, plasticity allows for a degree of flexibility in responding to the situation. For the purpose of keeping it simple, here we will focus the discussion on immune plasticity on that of the celebrities (in my opinion) – T cells. These lymphocytes have multiple intermediate phenotypes which were previously thought to be polarized subsets.
Understanding Plasticity
To discuss plasticity in broader terms, a discussion of the state of the field by Mills et al. in EMBO nicely illustrates cellular differentiation and plasticity as balls within a groove. The most relevant type of plasticity in immunity is trans differentiation, where cells acquire new cell identity without reversion to an immature state. To properly define plasticity, Mills et al. suggested that certain concepts need to be clarified:
- Is it trans differentiation or re-differentiation?
- If the cell divides during the transition to acquire new functions, is it still trans differentiation?
- Are these cells acquiring new phenotypes or are they a completely new subset of cells?
- How many new features does it have to adopt before defining as being a different cell type?
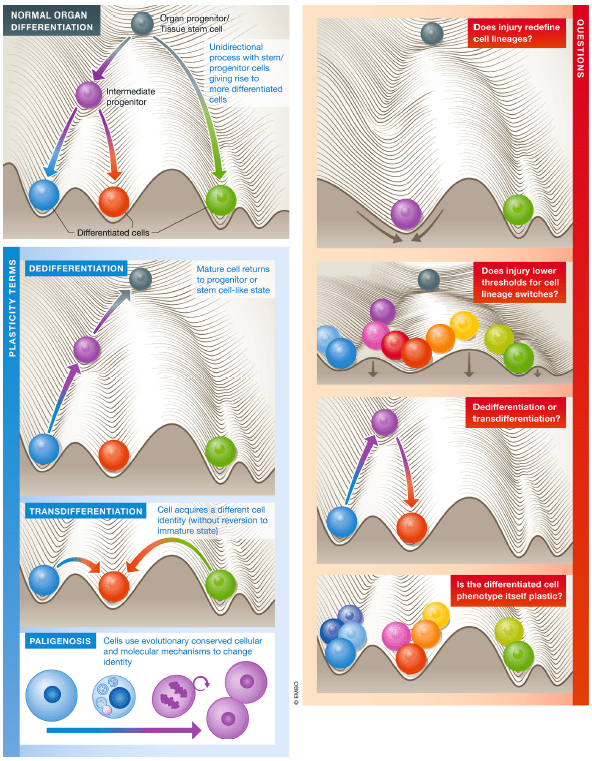
While some of these questions remain for the most part unanswered due to the technical limitations of observing dynamic processes in- vivo, immunologists have continued to move forward with categorizing new subsets of T cells and deciphering the mechanisms behind these intermediate phenotypes. A review by DuPage and Bluestone in 2016 comprehensively summarizes the different mechanisms of T cell plasticity as we understand it so far, where immune plasticity generally depends on the milieu of the stimulus present (strength and composition of cytokine/TCR stimulus).
An illustration of different types of plasticities and unanswered questions in a tissue differentiation landscape envisioned by Waddington. Grooves indicating the tissue environment, whereas bottom of the grooves indicating terminal differentiation. Figure taken from Mills et al. (2019).
Plasticity – Beneficial or Detrimental?
Now, is plasticity just a remnant of evolution, where its vestigial function remains to be selected out? How crucial is it to the daily functioning of our immune system? Turns out, these cells are just as functional as their single-minded counterparts depending on the situation – raising the question of ‘if we only had these plastic cells, wouldn’t we be able to cope with every situation appropriately?’ The keyword here is ‘appropriate’, since our immune system, while extremely impressive, also has its flaws. To paraphrase a certain superhero: ‘with great flexibility comes frequent miscommunication’. T cell plasticity is also involved in a medley of immune-driven diseases – ranging from Th1/Th17 cells in multiple sclerosis to Th17/Tregs in colorectal cancer.
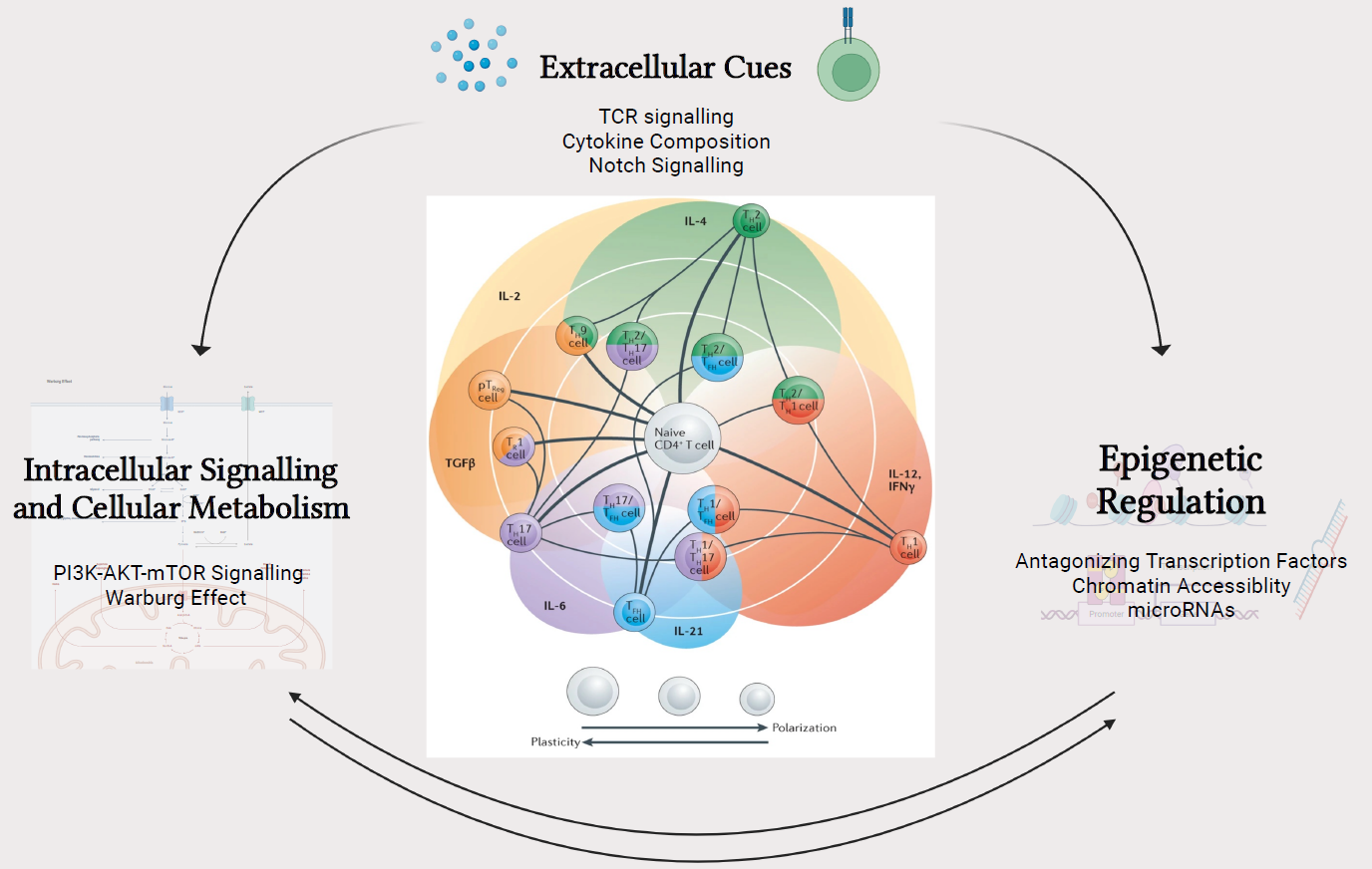
The regulatory triad of regulating cell plasticity. Figure adapted from DuPage and Bluestone (2016). Created with Biorender.com.
Nevertheless, having flexible cells makes extraordinary evolutionary sense considering the breadth and evasiveness of the pathogens our body is exposed to. As pathogens are able to transition from one microenvironmental niche to another (i.e extracellular to intracellular in certain parasites), our immune system also needs to be able to effectively transition from one effector response to another to not lose ground in this race. Therefore, it is still extremely beneficial to maintain this flexibility afforded to us by plasticity. In the end, similar to many other facets in immunology, balance and appropriate regulation of this flexibility has to be maintained to keep the system running smoothly. To that extent, to understand how to manipulate this inherent flexibility in these cells, the mechanisms and concepts behind plasticity has been a topic of interest over the past two decades and is still under research.
The Way Forward
One of the ways we can discover new phenotypes is through in-silico predictions with mathematical modelling based of previous experimental data, followed by in-vivo validation using fate-mapping techniques and new single cell technologies such as scRNA-sequencing and mass cytometry (CyTOF) available for large scale immunophenotyping. So, we are getting there! While it can be slightly messy with the current state of cell phenotypes, as long as the phenotype of any cell associated with high plasticity is properly defined, it would still allow us to define its relevance in a disease regardless of its stability as a cell sublineage. A small dose of organized chaos has never hurt anybody.
Main Takeaways:
- Immune plasticity may complicate categorization of cells and questions the concept of immune cell identity.
- With the rise of single cell analysis techniques, large scale immunophenotyping would allow us to better understand these plastic populations by giving insights into the immune landscape as a complex whole instead of a biased attempt at simplifying immune populations.
- T cell plasticity can be exploited and harnessed for future therapies in different diseases.
The SYIS does not guarantee the accuracy of the content published in this blog. The content does not necessarily reflect the opinion or views of the SYIS.

Leave a comment